M-Star CFD software is cited in scientific journal for its model validation of Novell Lattice Boltzmann Large Eddy Simulations in biopharma.
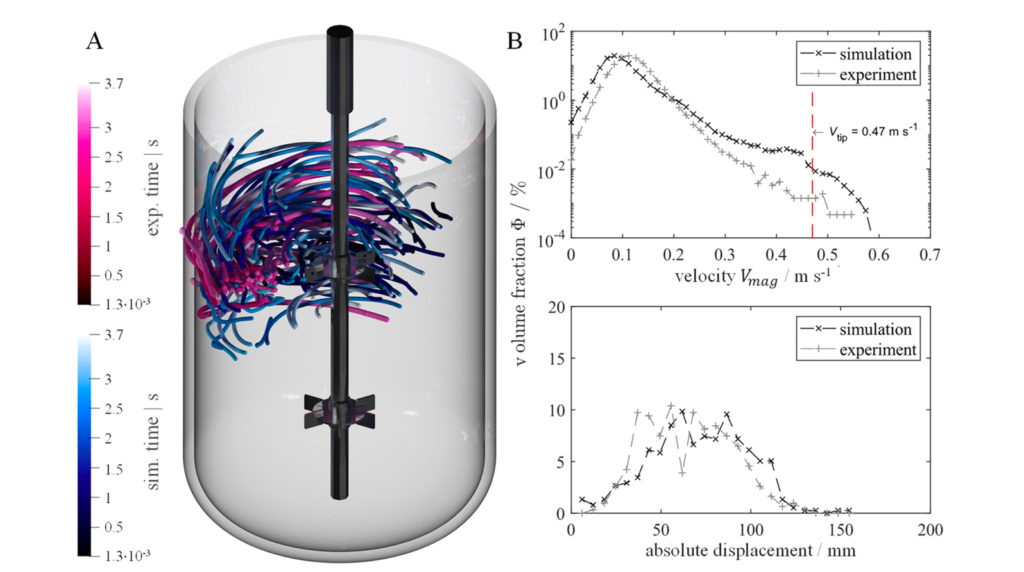
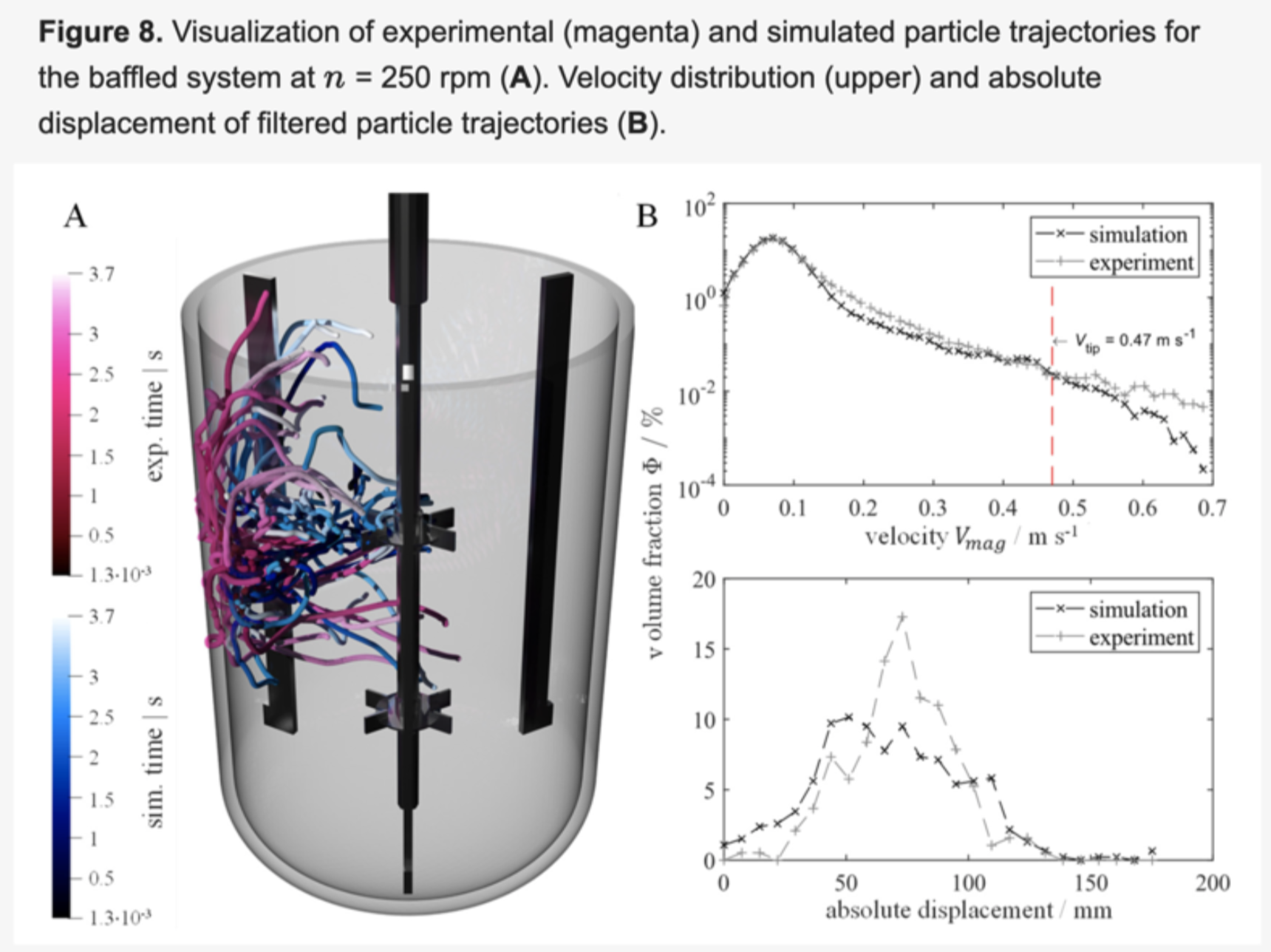
M-Star CFD software is cited in the journal Processes. The article records the first time the Lattice Boltzmann Method was proven for Large Eddy Simulations and supports biopharma taking biologic processes from lab-scale to production-scale with greater speed and fidelity.
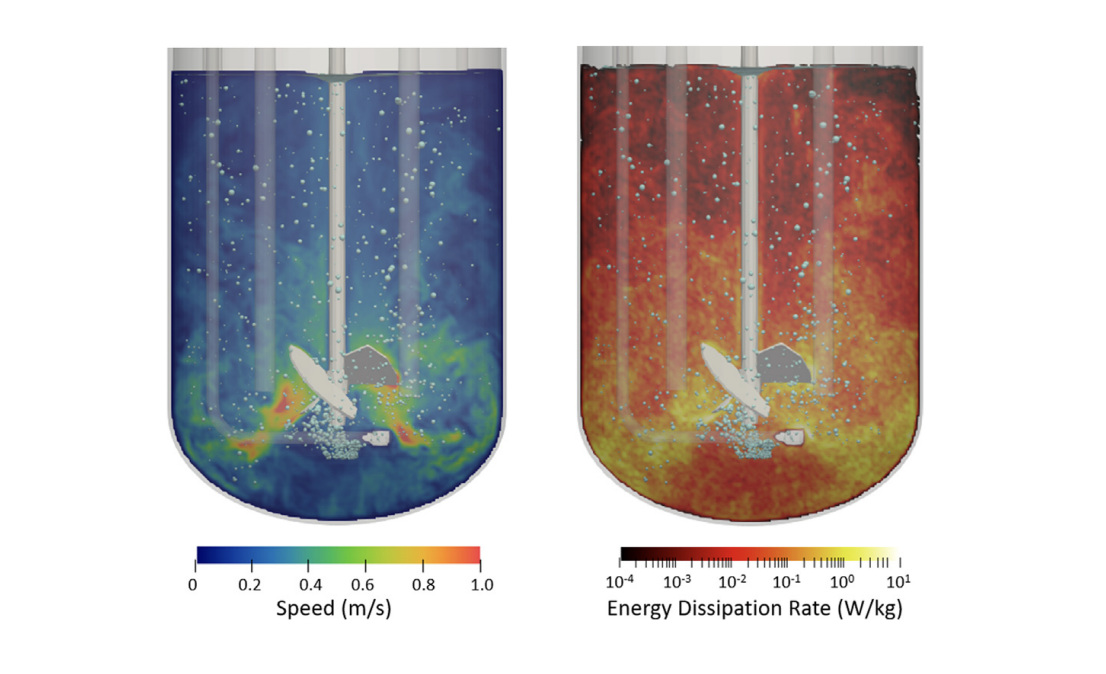
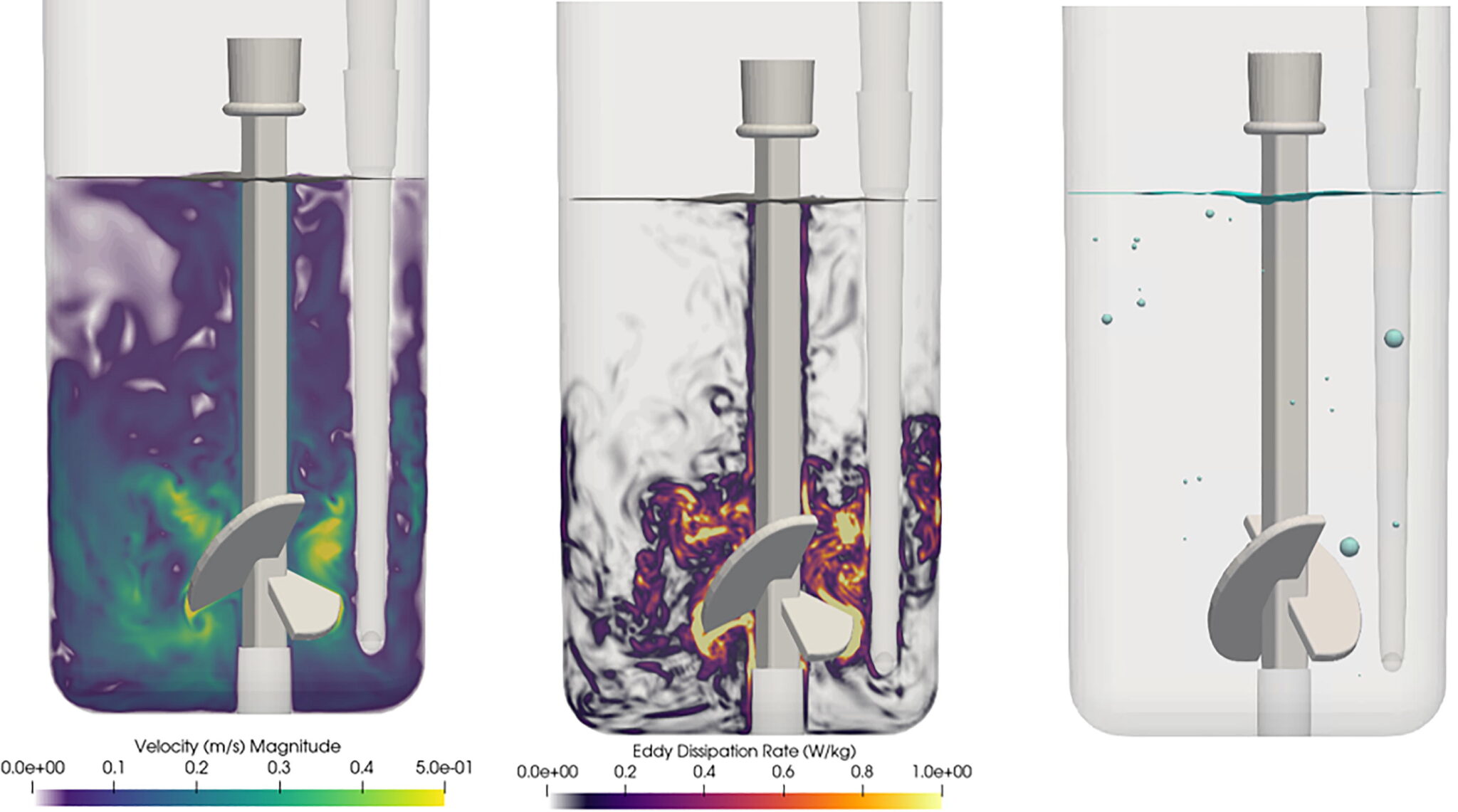
M-Star CFD handles fluid dynamic simulations with millions of individually tracked bubbles, allowing biopharma companies to model complex bioreactor physics problems—including mass transfer and multiphase fluid flow—accurately and in minutes.
“M-Star CFD creates a ‘digital twin’ with computational fluid dynamics to validate things other CFD tools cannot,” says John Thomas, PhD, president of M-Star. “The article published in Processes shows that M-Star CFD’s simulation results are indistinguishable from what can be measured in a lab. M-Star CFD has helped advance COVID-19 vaccines and many other important biopharma products.”
 Explore the Scientific R&D Software
Explore the Scientific R&D Software